
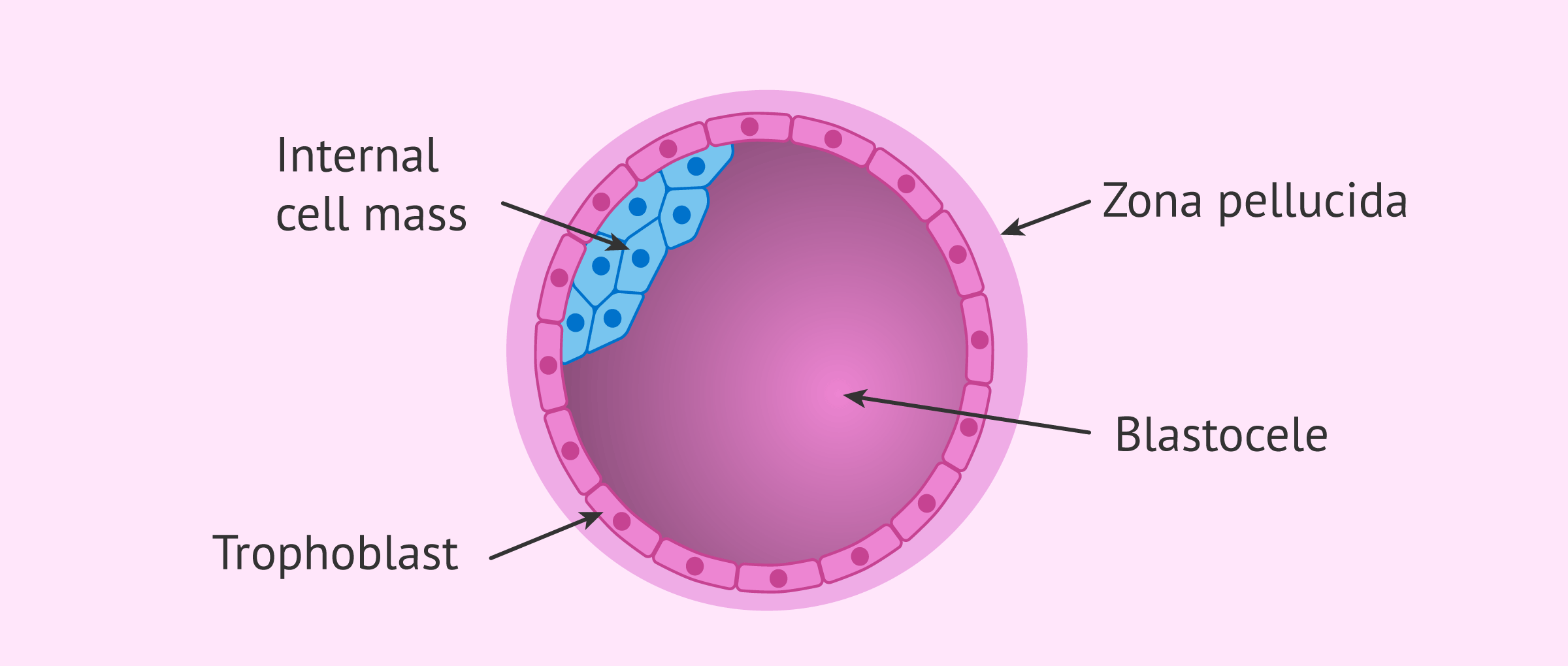
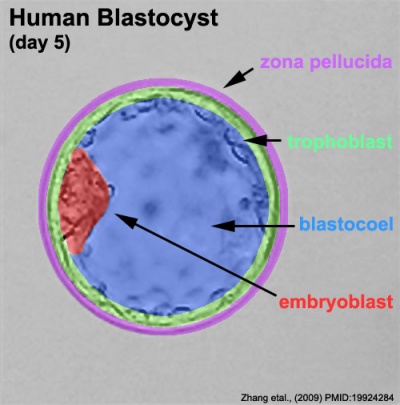
Blastocyst the BLASTULA of mammals, differing from that of lower vertebrates in consisting of an outer wall of cells, the TROPHOBLAST, by which the embryo implants in the uterus, and in having an inner cell mass, the embryonic disc, from which the embryo is formed.
Fertilization in humans. The sperm and ovum unite through fertilization, creating a conceptus that (over the course of 8-9 days) will implant in the uterine wall, where it will reside over the course of nine months.In humans, implantation is the stage of at which the embryo adheres to the wall of the. At this stage of, the is called a.
It is by this adhesion that the embryo receives oxygen and nutrients from the mother to be able to grow.In humans, implantation of a is most likely to occur around nine days after; however, this can range between six and 12 days. Contents.Implantation window The reception-ready phase of the of the uterus is usually termed the 'implantation window' and lasts about 4 days. The implantation window occurs around 6 days after the peak in levels.
With some disparity between sources, it has been stated to occur from 7 days after ovulation until 9 days after ovulation, or days 6-10 postovulation. On average, it occurs during the 20th to the 23rd day after the.The implantation window is characterized by changes to the endometrium cells, which aid in the absorption of the uterine fluid. These changes are collectively known as and bring the nearer to the endometrium and immobilize it.
During this stage the blastocyst can still be eliminated by being flushed out of the uterus. Scientists have hypothesized that the hormones cause a swelling that fills the flattened out uterine cavity just prior to this stage, which may also help press the blastocyst against the endometrium.
The implantation window may also be initiated by other preparations in the endometrium of the uterus, both structurally and in the composition of its secretions.Adaptation of uterus To enable implantation, the uterus goes through changes in order to be able to receive the conceptus.Predecidualization The endometrium increases thickness, becomes and its glands grow to be tortuous and boosted in their secretions. These changes reach their maximum about 7 days after.Furthermore, the surface of the endometrium produces a kind of rounded cells, which cover the whole area toward the uterine cavity.
This happens about 9 to 10 days after ovulation. These cells are called, which emphasises that the whole layer of them is shed off in every if no pregnancy occurs, just as leaves of. The uterine glands, on the other hand, decrease in activity and degenerate around 8 to 9 days after ovulation in absence of pregnancy.The originate from the stromal cells that are always present in the endometrium. However, the decidual cells make up a new layer, the. The rest of the endometrium, in addition, expresses differences between the luminal and the basal sides.
The luminal cells form the of the endometrium, in contrast to the basalolateral, which consists of the rather spongy stromal cells. Decidualization Decidualization succeeds predecidualization if pregnancy occurs. This is an expansion of it, further developing the uterine glands, the zona compacta and the epithelium of decidual cells lining it. The decidual cells become filled with lipids and glycogen and take the polyhedral shape characteristic for decidual cells.Trigger It is likely that the blastocyst itself makes the main contribution to this additional growing and sustaining of the decidua. An indication of this is that decidualization occurs at a higher degree in conception cycles than in nonconception cycles.
Furthermore, similar changes are observed when giving stimuli mimicking the natural invasion of the embryo.The embryo releases serine proteases which causes the epithelial cell membrane to depolarize and activates the epithelial Na+ channel. This triggers a Ca2+ influx and phosphorylation of CREB. Phosphorylation of CREB upregulates the expression of COX-2, which leads to the release of prostaglandin E2 (PGE2) from epithelial cells. PGE2 acts on the stroma cells activating cAMP-related pathways in stromal cell leading to decidualization. Parts of decidua The decidua can be organized into separate sections, although they have the same composition.
Decidua basalis - This is the part of the decidua which is located basalolateral to the embryo after implantation. Decidua capsularis - Decidua capsularis grows over the embryo on the luminal side, enclosing it into the endometrium. It surrounds the embryo together with decidua basalis. Decidua parietalis - All other decidua on the uterine surface belongs to decidua parietalis.Decidua throughout pregnancy After implantation the decidua remains, at least through the first trimester.
However, its most prominent time is during the early stages of pregnancy, during implantation. Its function as a surrounding tissue is replaced by the definitive. However, some elements of the decidualization remain throughout pregnancy.The compacta and spongiosa layers are still observable beneath the decidua in pregnancy. The glands of the spongiosa layer continue to secrete during the first trimester, when they degenerate. However, before that disappearance, some glands secrete unequally much.
This phenomenon of hypersecretion is called the, after the pathologist.Pinopodes. Main article:Pinopodes are small, finger-like protrusions from the endometrium. They appear between day 19 and day 21 of. This corresponds to a of approximately five to seven days, which corresponds well with the time of implantation. They only persist for two to three days.
The development of them is enhanced by but inhibited by.Function in implantation Pinopodes uterine fluid and macromolecules in it. By doing so, the volume of the uterus decreases, taking the walls closer to the embryoblast floating in it. Thus, the period of active pinocytes might also limit the implantation window. Function during implantation Pinopodes continue to absorb fluid, and removes most of it during the early stages of implantation.Adaptation of secretions proteins, glycoproteins and peptidessecreted by the endometrial glands-associated:-Others:-1(PP14) or glycodelinPregnancy-associated endometrialalpha-2-globulin (PAPP-A)(SRP-27)Leu-DiProgesterone-dependentNot only the lining of the uterus transforms, but in addition, the secretion from its epithelial glands changes.
This change is induced by increased levels of from the. The target of the secretions is the embryoblast, and has several functions on it.Nourishment The embryoblast spends approximately 72 hours in the uterine cavity before implanting. In that time, it cannot receive nourishment directly from the blood of the mother, and must rely on secreted nutrients into the uterine cavity, e.g. Iron and fat-soluble vitamins. Growth and implantation In addition to nourishment, the endometrium secretes several -dependent proteins, important for growth and implantation.
And steroids are also secreted. Implantation is further facilitated by synthesis of substances, and surface for the matrix substances.Mechanism Implantation is initiated when the blastocyst comes into contact with the uterine wall.Zona hatching. Main article:To be able to perform implantation, the blastocyst first needs to get rid of its. This process can be called 'hatching'.Factors Lytic factors in the uterine cavity, as well as factors from the blastocyst itself are essential for this process.
Mechanisms in the latter are indicated by that the zona pellucida remains intact if an unfertilized egg is placed in the uterus under the same conditions. A substance probably involved is., the plasmin precursor, is found in the uterine cavity, and blastocyst factors contribute to its conversion to active plasmin. This hypothesis is supported by lytic effects by plasmin. Furthermore, plasmin inhibitors also inhibit the entire zona hatching in rat experiments. Apposition The very first, albeit loose, connection between the blastocyst and the endometrium is called the apposition. Location On the endometrium, the apposition is usually made where there is a small crypt in it, perhaps because it increases the area of contact with the rather spherical blastocyst.On the blastocyst, on the other hand, it occurs at a location where there has been enough lysis of the zona pellucida to have created a rupture to enable direct contact between the underlying and the decidua of the endometrium. However, ultimately, the, inside the layer, is aligned closest to the decidua.
Nevertheless, the apposition on the blastocyst is not dependent on if it is on the same side of the blastocyst as the inner cell mass. Rather, the inner cell mass rotates inside the trophoblast to align to the apposition. In short, the entire surface of the blastocyst has a potential to form the apposition to the decidua.Molecular Mechanism The identity of the molecules on the trophoblast and the endometrial epithelia that mediate the initial interaction between the two remain unidentified. However, a number of research groups have proposed that, a member of the family of glycosylated proteins, is involved. MUC1 is a transmembrane expressed at the apical surface of endometrial epithelial cells during the window of implantation in humans and has been shown to be differentially expressed between fertile and infertile subjects during this time. MUC1 displays carbohydrate moieties on its extracellular domain that are ligands of, a protein expressed on the surface of trophoblast cells.
An in vitro model of implantation developed by Genbacev et al., gave evidence to support the hypothesis that L-selectin mediates apposition of the blastocyst to the uterine epithelium by interacting with its ligands. Adhesion Adhesion is a much stronger attachment to the endometrium than the loose apposition.The trophoblasts adhere by penetrating the endometrium, with protrusions of trophoblast cells.This adhering activity is by microvilli that are on the trophoblast. The trophoblast have binding fiber connections, laminin, collagen type IV, and integrins that assist in this adhesion processMUC16 is a transmembrane mucin expressed at the apical surface of uterine epithelia. This mucin prevents the blastocyst from implanting in an undesired located on the epithelium. Thus, MUC16 inhibits cell-cell adhesion. “Removal of this mucin during formation of uterodomes (bulbous projections from the apical surface of the epithelium that are often found during the implantation period) facilitates trophoblast adhesion in vitro”. Communication There is massive communication between the blastocyst and the endometrium at this stage.
The blastocyst signals to the endometrium to adapt further to its presence, e.g. By changes in the of decidual cells. This, in turn, dislodges the decidual cells from their connection to the underlying, which enables the blastocyst to perform the succeeding invasion.This communication is conveyed by -interactions, both integrin-matrix and proteoglycan ones.Proteoglycan Receptors Another ligand-receptor system involved in adhesion is proteoglycan receptors, found on the surface of the decidua of the uterus. Their counterparts, the proteoglycans, are found around the trophoblast cells of the blastocyst. This ligand-receptor system also is present just at the implantation window. Invasion Invasion is an even further establishment of the blastocyst in the endometrium.Syncytiotrophoblasts The protrusions of trophoblast cells that adhere into the endometrium continue to proliferate and penetrate into the endometrium. As these trophoblast cells penetrate, they differentiate to become a new type of cells,.
The prefix syn- refers to the transformation that occurs as the boundaries between these cells disappear to form a single mass of many. The rest of the trophoblasts, surrounding the inner cell mass, are hereafter called. Syncytiotrophoblast is not determined as a cell type, rather is a multinucleated tissueInvasion continues with the syncytiotrophoblasts reaching the basal membrane beneath the decidual cells, penetrating it and further invading into the uterine stroma.
Finally, the whole embryo is embedded in the endometrium. Eventually, the syncytiotrophoblasts come into contact with maternal blood and form. This is the initiation of forming the.The penetration of the trophoblast to the endometrium is demonstrated through metalloproteinase MMP-2 and MMP-9 Syncytiotrophoblast invade the uterus attempting to reach maternal blood supply, for setting up the foundation for fetal blood flow Extravillous trophoblasts Extravillous trophoblasts are cells from the invading villi that migrate into the myometrium of the mother’s uterus. These cells remodel the spiral arteries to improve and secure maternal blood flow to the growing embryo. There is also evidence that this process occurs with the uterine veins. Stabilizing them to improve drainage of fetal blood and metabolic wastes.
Trophoblasts have also been documented to migrate into the mother and have been found in various tissues. Due to this trophoblasts have been implicated in a phenomenon known as “Fetomaternal Microchimerism” where fetal cells establish cell lines in maternal tissues. Secretions The blastocyst secretes factors for a multitude of purposes during invasion. It secretes several factors, targeting itself and stimulating it to further invade the endometrium. Furthermore, secretions loosen decidual cells from each other, prevent the embryo from being rejected by the mother, trigger the final decidualization and prevent menstruation.Autocrine is an autocrine growth factor for the blastocyst., on the other hand, stimulates the invasiveness of it.Dislodging The syncytiotrophoblasts dislodges decidual cells in their way, both by degradation of linking the decidual cells together as well as degradation of the extracellular matrix between them.Cell adhesion molecules are degraded by syncytiotrophoblast secretion of. This inhibits the expression of. Cadherins are cell adhesion molecules, and beta-catenin helps to anchor them to the cell membrane.
Inhibited expression of these molecules thus loosens the connection between decidual cells, permitting the syncytotrophoblasts and the whole embryo with them to invade into the endometrium.The extracellular matrix is degraded. Examples of such metalloproteinases are,.
These collagenases digest, Type-VII collagen and Type-X collagen. The gelatinases exist in two forms; one digesting and one digesting. Immunosuppressive The embryo differs from the cells of the mother, and would be rejected as a parasite by the of the mother if it didn't secrete agents. Such agents are, 2, -alpha, -alpha, and.Decidualization Factors from the blastocyst also trigger the final formation of decidual cells into their proper form. In contrast, some decidual cells in the proximity of the blastocyst degenerate, providing nutrients for it. Prevention of menstruation (hCG) not only acts as an immunosuppressive, but also 'notifies' the mother's body that she is, preventing menstruation by sustaining the function of the.Other factors Other factors secreted by the blastocyst are;.
Embryo-derived. as well as its inhibitors. β1-.Failure Implantation failure is considered to be caused by inadequate uterine receptivity in two-thirds of cases, and by problems with the embryo itself in the other third.Inadequate uterine receptivity may be caused by abnormal and hormonal signaling as well as. Recurrent implantation failure is a cause of.
Therefore, can be improved by optimizing endometrial receptivity for implantation. Evaluation of implantation markers may help to predict pregnancy outcome and detect occult implantation deficiency.is the administration of medication, generally, for the purpose of increasing the success rate of implantation and early, thereby complementing the function of the.In women with more than 3 implantation failures in, a review of several small estimated that the use of adjunct (LMWH) improves by approximately 80%.
See also.References. Wilcox AJ, Baird DD, Weinberg CR (1999). 'Time of implantation of the Conceptus and loss of pregnancy'. New England Journal of Medicine. 340 (23): 1796–1799. Xiao, Y.; Sun, X.; Yang, X.; Zhang, J.; Xue, Q.; Cai, B.; Zhou, Y. 'Leukemia inhibitory factor is dysregulated in the endometrium and uterine flushing fluid of patients with adenomyosis during implantation window'.
Fertility and Sterility. 94 (1): 85–89. Aboubakr M.
Elnashar, Gamal I. Middle East Fertility Society Journal, Vol.
10-24. from embryology.ch at by the universities of Fribourg, Lausanne and Bern (Switzerland). Retrieved May, 2012. Human Embryology. Online course in embryology for medicine students developed by the universities of Fribourg, Lausanne and Bern (Switzerland) with the support of the Swiss Virtual Campus.
Retrieved 6 December 2011. ^; (2004). Medical Physiology: A Cellular And Molecular Approaoch.:.
Ruan, Y. H., Liu, X., Zhang, R., Tsang, L.
Activation of the epithelial Na channel triggers prostaglandin E2 release and production required for embryo implantation. Nature Medicine, 18(7), 1112–1117. Doi: 10.1038/nm.2771. ^ Margarit, L.; Taylor, A.; Roberts, M. H.; Hopkins, L.; Davies, C.; Brenton, A.
G.; Conlan, R. S.; Bunkheila, A.; Joels, L.; White, J. O.; Gonzalez, D.
'MUC1 as a Discriminator between Endometrium from Fertile and Infertile Patients with PCOS and Endometriosis'. The Journal of Clinical Endocrinology & Metabolism.
95 (12): 5320–5329. Carson, D. D.; et al., (2006). 'MUC1 is a scaffold for selectin ligands in the human uterus'. 11 (1): 2903. Genbacev, O. D.; et al., (2003).
'Trophoblast L-selectin-mediated adhesion at the maternal-fetal interface'. 299 (5605): 405–8. Singh, H., & Aplin, J. Adhesion molecules in endometrial epithelium: tissue integrity and embryo implantation. Journal of anatomy, 215(1), 3–13.
Doi:10.1111/j.1469-7580.2008.01034.x. Gipson IK, Blalock T, Tisdale A, et al. (2008) MUC16 is lost from the uterodome (pinopode) surface of the receptive human endometrium: in vitro evidence that MUC16 is a barrier to trophoblast adherence. Biol Reprod 78, 134– 142.
Gardiner, C., Tannetta, D., Simms, C., Harrison, P., Redman, C., Sargent, I., & Milstone, D. Syncytiotrophoblast Microvesicles Released from Pre-Eclampsia Placentae Exhibit Increased Tissue Factor Activity (Tissue Factor in Pre-Eclampsia).
PLoS ONE, 6(10), e26313. Zhu, J. Regulation of trophoblast invasion: the role of matrix metalloproteinases.
Reviews in obstetrics & gynecology, 5(3-4), e137–e143. Carter, A. M., Enders, A. C., & Pijnenborg, R. The role of invasive trophoblast in implantation and placentation of primates.
Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 370(1663), 20140070. Doi:10.1098/rstb.2014.0070. Moser, G., Weiss, G., Sundl, M., Gauster, M., Siwetz, M., Lang-Olip, I., & Huppertz, B. Extravillous trophoblasts invade more than uterine arteries: evidence for the invasion of uterine veins. Histochemistry and Cell Biology, 147(3), 353–366.
Doi: 10.1007/s00418-016-1509-5. Dawe, G. W., & Xiao, Z.-C. Cell Migration from Baby to Mother. Cell Adhesion & Migration, 1(1), 19–27. Doi: 10.4161/cam.1.1.4082.
Ai Z., Jing W., Fang L. 11 (1): e0147424. CS1 maint: multiple names: authors list. Melford, S. E.; Taylor, A. H.; Konje, J.
'Of mice and (wo)men: factors influencing successful implantation including endocannabinoids'. Human Reproduction Update. 20 (3): 415–428. ^ Cakmak, H.; Taylor, H.
Human Reproduction Update. 17 (2): 242–253.
Potdar, N.; Gelbaya, T. A.; Konje, J. C.; Nardo, L.
'Adjunct low-molecular-weight heparin to improve live birth rate after recurrent implantation failure: A systematic review and meta-analysis'. Human Reproduction Update.
19 (6): 674–684.Further reading.

